Research
Our general research interests lie in the development and application of optical spectroscopic and microscopic tools to address quantitatively chemical and biological questions at the molecular scale. Time-resolved fluorescence spectroscopy and single-molecule fluorescence methods including super-resolution microscopy are particularly useful tools in this context.
We aim at better understanding chemical and biological systems ranging from simple fluorescent probes and luminescent coordination complexes to soft matter (microgel particles, polymers) or complex biological systems (G protein-coupled receptors or other proteins). A particular emphasis is put on understanding in detail the photophysics of the fluorescent probes we use in order to be able to use them as quantitative sensors of their local environment.
Quantitative fluorescent sensors for single-molecule and super-resolution imaging
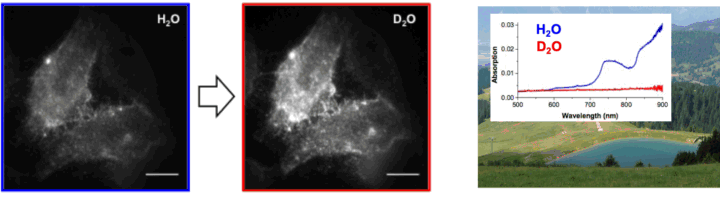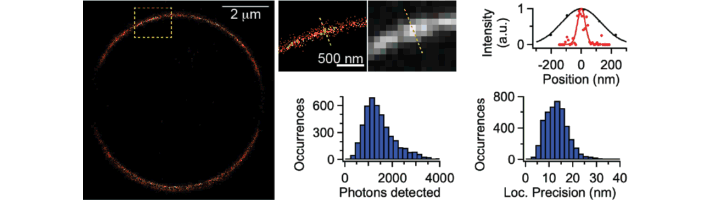 |
The emission properties of a fluorescent dye depend on the direct environment of the probe. Changes in the fluorescence intensity, spectrum, lifetime or polarization anisotropy can therefore directly report on the local surroundings of the fluorophore and this effect has been used to develop numerous qualitative fluorescence sensors. An understanding of the exact origin of the fluorescence modulation is however necessary to make use of this effect quantitatively and we are using our expertise in molecular photophysics to develop quantitative fluorescence sensors enabling to count water molecules surrounding the fluorophore or enabling to measure structural dynamics based on photoinduced electron transfer.
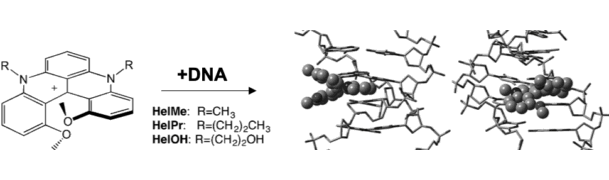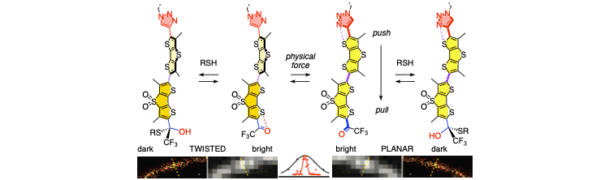
We are further developing ways to quantitatively image lateral forces in lipid membranes at nanometer resolution with mechanosensitive fluorescent probes.
For recent references, see publications 20, 23, 24, 26, 31, 42.
Protein dynamics and G protein-coupled receptors
 |
We have been developing and applying single-molecule methods to study the conformational dynamics and diversity of G protein-coupled receptors (GPCRs). GPCRs are key membrane proteins which mediate signalling across the cell surface and which are currently targeted by about one third of all available medicines. Rhodopsin, which enables vision, or the adrenergic receptors, which regulate our heart beat through the effect of e.g. adrenaline, are some commonly known GPCRs. A detailed comprehension of the molecular mechanisms underlying GPCR activation and function is still lacking, thereby preventing efficient rational drug design. We are tackling this issue by using cutting-edge biophysics tools such as live-cell fluorescence imaging, single-molecule microscopy and spectroscopy, super-resolution imaging, or single-particle tracking in addition to standard cellular and molecular biology tools.
For recent references, see publications 20, 25, 27, 29, 30, 35, 38.
Novel fluorescent probes
 |
By working with organic chemists, we are further interested in the development of novel fluorescent probes with unusual and interesting properties, especially those which enable direct affinity labelling of target molecules or target organelles, and those suitable for single-molecule based super-resolution microscopy.
For recent references, see publications 22 and 42.
Luminescent open-shell coordination complexes
 |
We have also been developing ways to investigate the luminescence properties of open-shell molecular systems for in-house collaborative projects. We have set up an experimental platform with which the luminescence of both organic and open-shell systems in the solid state or in solution phase can be studied without interruption from 260 to 1700 nm and within a temperature range from 10 to 400 K. Weakly absorbing metal centers can be efficiently directly excited thanks to 7 high-power laser lines covering the 720-980 nm spectral range, and both spectral and lifetime information can be obtained owing to versatile excitation and detection modalities. We have in particular been probing the mechanisms of light up-conversion in molecular erbium complexes.
For recent references, see publications 33, 34, 37, 40, 41, 43.
Spectroscopic techniques we use and develop in the lab
- Single-molecule fluorescence spectroscopy
- Single-molecule imaging, single-particle tracing
- Single-molecule based super-resolution microscopy (SMLM, dSTORM, PAINT)
- Fluorescence correlation spectroscopy (FCS)
- Time-resolved fluorescence spectroscopy
- Steady-state absorption and fluorescence spectroscopy
