Publication 106
- Enantioselective CpRu-Catalyzed Carroll Rearrangement. Metal Source Importance and Ligand Optimization
David Linder, Frédéric Buron, Samuel Constant, Jérôme Lacour
Eur. J. Org. Chem. 2008, 5778-5785
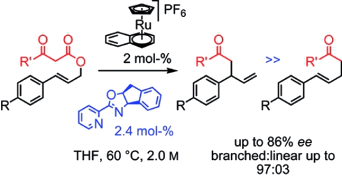
The addition of unstabilized carbonyl nucleophiles to unsymmetrical allyl-metal fragments still represents a challenge to generate stereogenic centers enantio- and regioselectively. In this context, the decarboxylative Carroll rearrangement of allyl β-keto esters is particularly interesting, since chiral γ,δ-unsaturated ketones are obtained. Herein, we show that CpRu half-sandwich complexes can, with selected enantiopure pyridine-monooxazoline ligands, catalyze this transformation and afford complete conversions along with good levels of regioselectivity and enantioselectivity. Even more challenging (electron-poor) substrates react (up to 86 % ee, branched/linear ratio ≥ 97:03). In addition, the use of an air-stable metal precursor, namely [CpRu(η6-naphthalene)][PF6], allows the reaction to be carried out reproducibly evenin non-anhydrous THF with a catalyst loading as low as 2 mol-%.
DOI : 10.1002/ejoc.200800854
archive ouverte unige:7952
