Publication 232
- Chemo- and Regioselective Multiple C(sp2)−H Insertions of Malonate Metal Carbenes for Late-Stage Functionalizations of Azahelicenes
Yana Nikolova, Bibiana Fabri, Pau Moneva Lorente, Alejandro Guarnieri-Ibáñez, Adiran de Aguirre, Yoshiki Soda, Gennaro Pescitelli, Francesco Zinna, Céline Besnard, Laure Guénée, Dimitri Moreau, Lorenzo Di Bari, Eric Bakker, Amalia Isabel Poblador Bahamonde, Jérome Lacour
Angew. Chem. Int. Ed. 2022, 61, e202210798
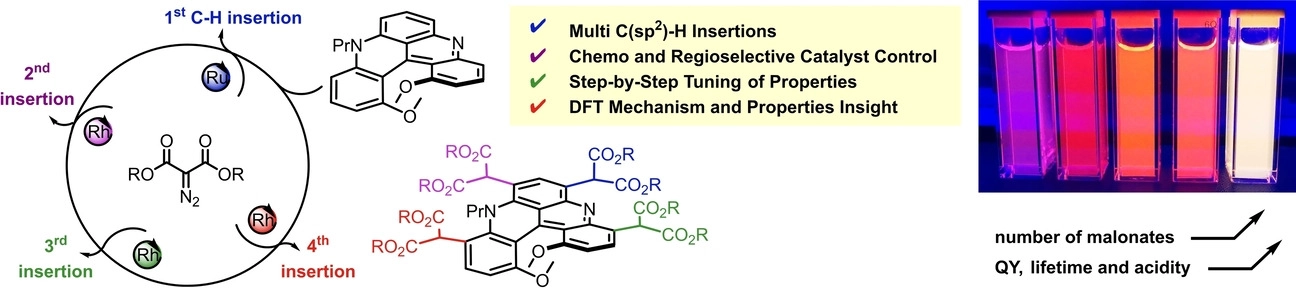
Chiral quinacridines react up to four times, step-by-step, with α-diazomalonates under Ru(II) and Rh(II) catalysis. By selecting the catalyst, [CpRu(CH3CN)3][PF6] (Cp = cyclopentadienyl) or Rh2(oct)4, chemo and regioselective insertions of derived metal carbenes are achieved in favor of mono- or bis-functionalized malonate derivatives respectively (r.r. > 49:1, up to 77% yield, 12 examples). This multi-introduction of malonate groups is particularly useful to tune optical and chemical properties such as absorption, emission or Brønsted acidity but also cellular bioimaging. Density-functional theory further elucidates the origin of the carbene insertion selectivity and also showcases the importance of conformations in the optical response.
DOI : 10.1002/anie.202210798
archive ouverte unige:164073
