Publication 14
- A Highly Effective Ruthenium System for the Catalyzed Dehydrogenative Cyclization of Amine-Boranes to Cyclic Boranes under Mild Conditions
Wallis, C. J.; Alcaraz, G.; Petit, A. S.; Poblador-Bahamonde, A. I.; Clot, E.; Bijani, C.; Vendier, L.; Sabo-Etienne, S.
Chem. Eur. J. 2015, 21, 13080-13090
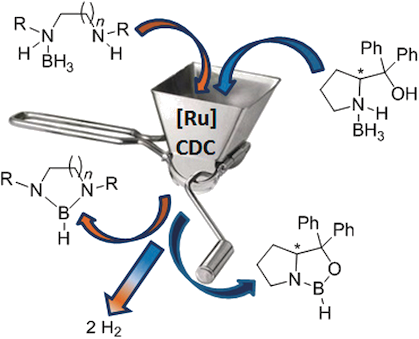
We recently disclosed a new ruthenium-catalyzed dehydrogenative cyclization process (CDC) of diamine–monoboranes leading to cyclic diaminoboranes. In the present study, the CDC reaction has been successfully extended to a larger number of diamine–monoboranes (4–7) and to one amine–borane alcohol precursor (8). The corresponding NB(H)N- and NB(H)O-containing cyclic diaminoboranes (12–15) and oxazaborolidine (16) were obtained in good to high yields. Multiple substitution patterns on the starting amine–borane substrates were evaluated and the reaction was also performed with chiral substrates. Efforts have been spent to understand the mechanism of the ruthenium CDC process. In addition to a computational approach, a strategy enabling the kinetic discrimination on successive events of the catalytic process leading to the formation of the NB(H)N linkage was performed on the six-carbon chain diamine–monoborane 21 and completed with a 15N?NMR study. The long-life bis-σ-borane ruthenium intermediate 23 possessing a reactive NHMe ending was characterized in situ and proved to catalyze the dehydrogenative cyclization of 1, ascertaining that bis σ-borane ruthenium complexes are key intermediates in the CDC process.
DOI : 10.1002/chem.201501569
archive ouverte unige:91983
