Recherches (en anglais)
ISOMERIZATIONS & REMOTE FUNCTIONALIZATIONS:
moving C=C bonds over short or long distances
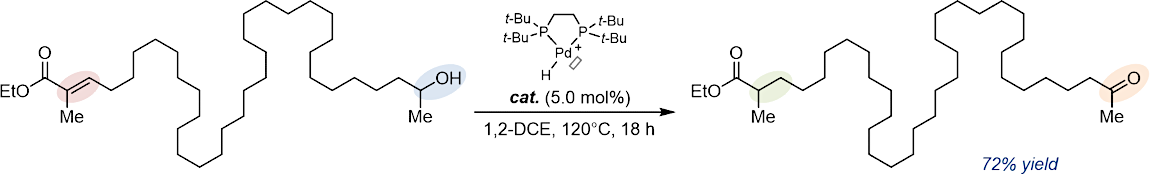
Transition metal-catalyzed isomerizations of alkenes not only offer the inherent advantages of atom-, step- and redox-economy but also enable the preparation of value-added products that would be difficult to access by conventional methods.
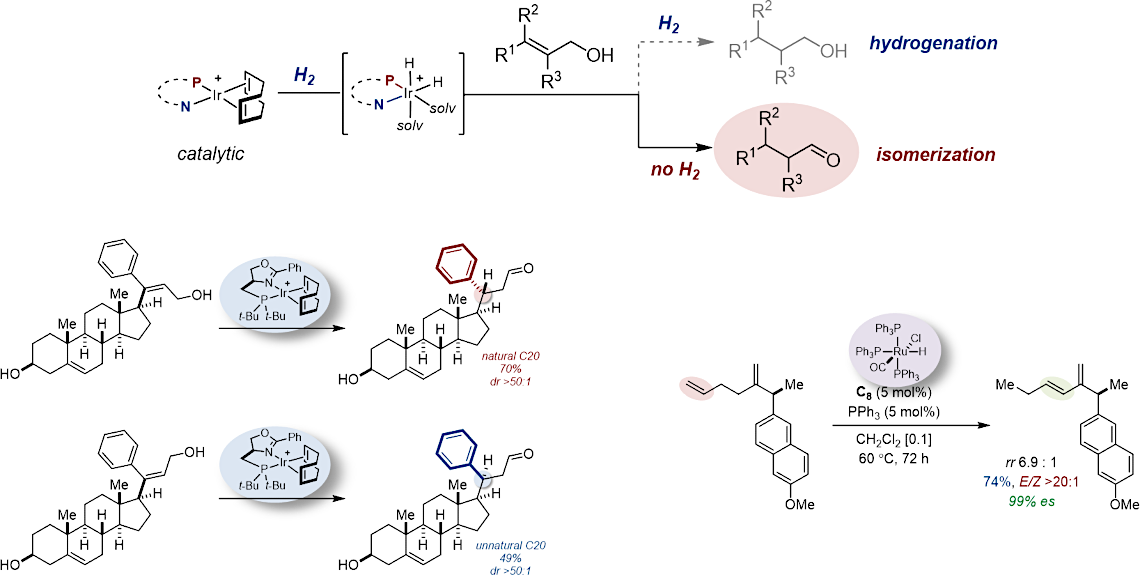
The remote functionalization of an organic molecule consists of interconverting two distant functional groups by a relay process either (i) through space or (ii) across the constitutional skeleton. The former usually requires the elaboration, installation, and subsequent removal of often complex directing templates to achieve the desired distant functionalization. The latter is typically accomplished by alkene migration along a hydrocarbon chain and driven by a thermodynamically favorable termination process. This enabling strategy has gained increased momentum over the past few years, leading to the development of particularly innovative transformations.